Discover the benefit of AJOVY® (fremanezumab) in preventive migraine treatment
Intended for UK Healthcare Professionals. Prescribing Information and Adverse Events reporting can be accessed in the footer of the site.
Migraine Space

What is AJOVY®?
AJOVY® is indicated for prophylaxis of migraine in adults who have at least four migraine days per month.¹
AJOVY® is a monoclonal antibody (mAb) that selectively binds to calcitonin gene-related peptide (CGRP), a neuropeptide elevated during migraines, preventing it from binding to the CGRP receptor.¹
By blocking the activation of the CGRP receptor, AJOVY® disrupts the chain of events that trigger migraines, ultimately reducing the frequency and severity of migraine attacks.1-3
AJOVY® is the only anti-CGRP monoclonal antibody approved for monthly and quarterly dosing.1
AJOVY® is available as a 225mg single dose injection in a pre-filled pen and as a pre-filled syringe.1


Effective option for individuals with migraine who have had previous exposure to other CGRP pathway antibodies4-6

Demonstrated efficacy and tolerability profile in patients with migraine and major depressive disorder7*

Generally well tolerated in the real world4,8-10

Demonstrated sustained reductions in MMDs over a 24-month period4,8
How does AJOVY® work?
CGRP is involved in several pathophysiological processes, including dilation of cerebral and dural blood vessels, modulating release of inflammatory mediators from mast cells, and transmission of sensory signals within the trigeminovascular system.11,12 Activation of trigeminal nerves leads to the release of CGRP and other neuropeptides, contributing to neurogenic inflammation, vasodilation of meningeal vessels, and sensitisation of peripheral trigeminovascular neurons, which are implicated in migraine pathophysiology.11,12
Despite CGRP being established as a key mediator capable of triggering migraine attacks, the precise sequence of events, relative contribution of individual processes, and finer mechanistic details remain incompletely understood.1,11,12
AJOVY® is a CGRP ligand binder, which means it selectively binds to CGRP and prevents it from activating the CGRP receptor1. Inhibition of CGRP-induced signaling thereby reduces the downstream cascade of events within trigeminovascular pathways involved in the development and maintenance of migraine.1,11,12
Play the video to learn more about how AJOVY® selectively binds to CGRP.
AJOVY® efficacy
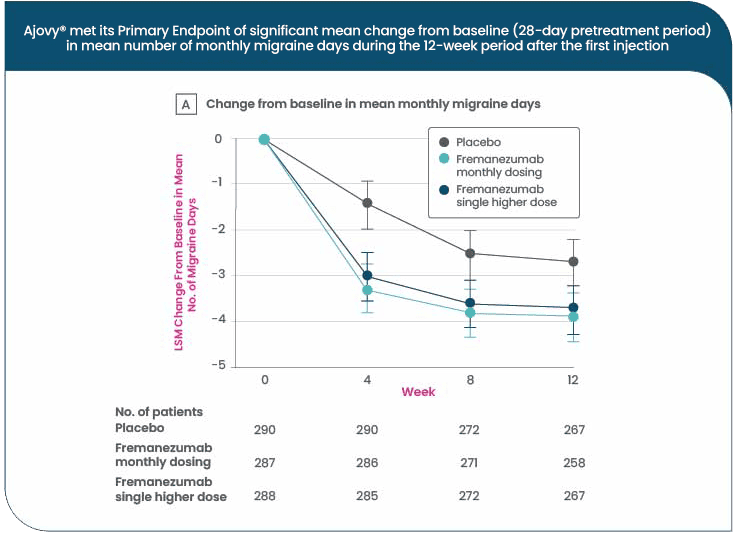
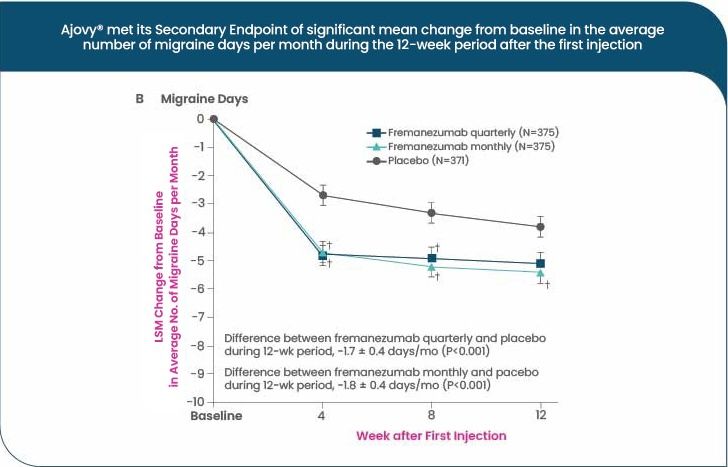
Statistically significant reductions in MMDs were demonstrated for both monthly and quarterly AJOVY® dosing regimens in pivotal Phase III randomised controlled trials, over three months13,14
AJOVY® in Episodic Migraine (EM)
Monthly average number of migraine days were reduced from Baseline by 3.7 days with AJOVY® monthly, 3.4 days with AJOVY® quarterly, and 2.2 days with placebo (primary endpoint, n=865; p<0.001)13
AJOVY® in Chronic Migraine (CM)
Monthly average number of headache days** were reduced from baseline by 4.6 days with AJOVY® monthly, 4.3 days with AJOVY® quarterly, and 2.5 days with placebo (primary endpoint, n=1121; p<0.001)14
Change in MMD was a secondary endpoint
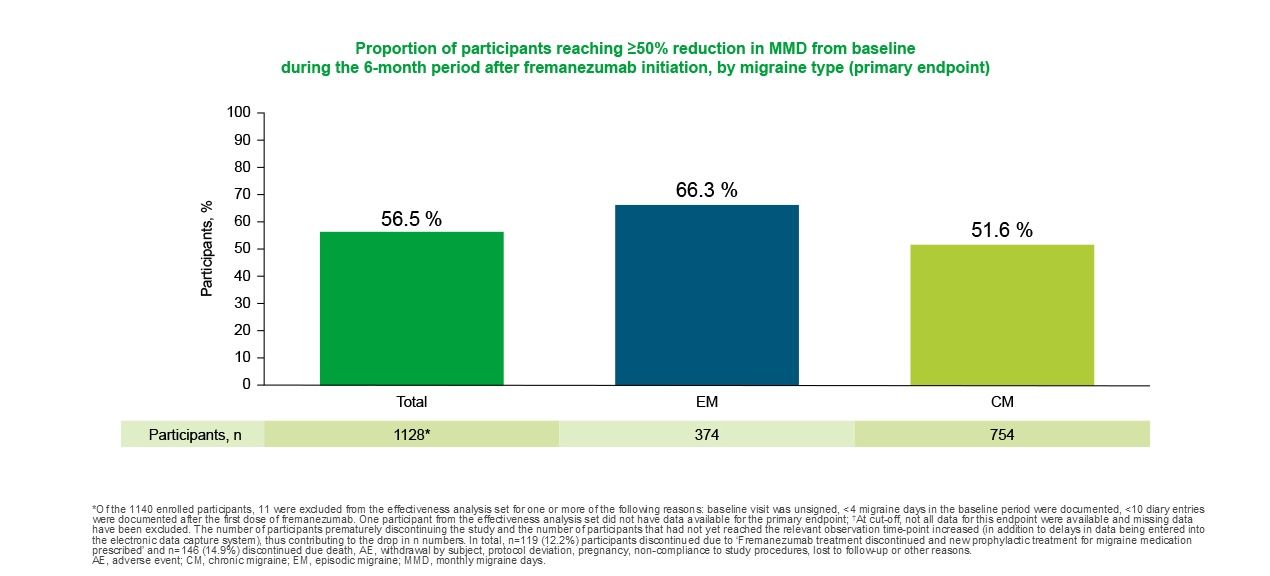
AJOVY® is supported by a wealth of clinical trial data and a growing body of real-world data on effectiveness, tolerability profile and adherence across a broad migraine patient population1,3-10
PEARL study
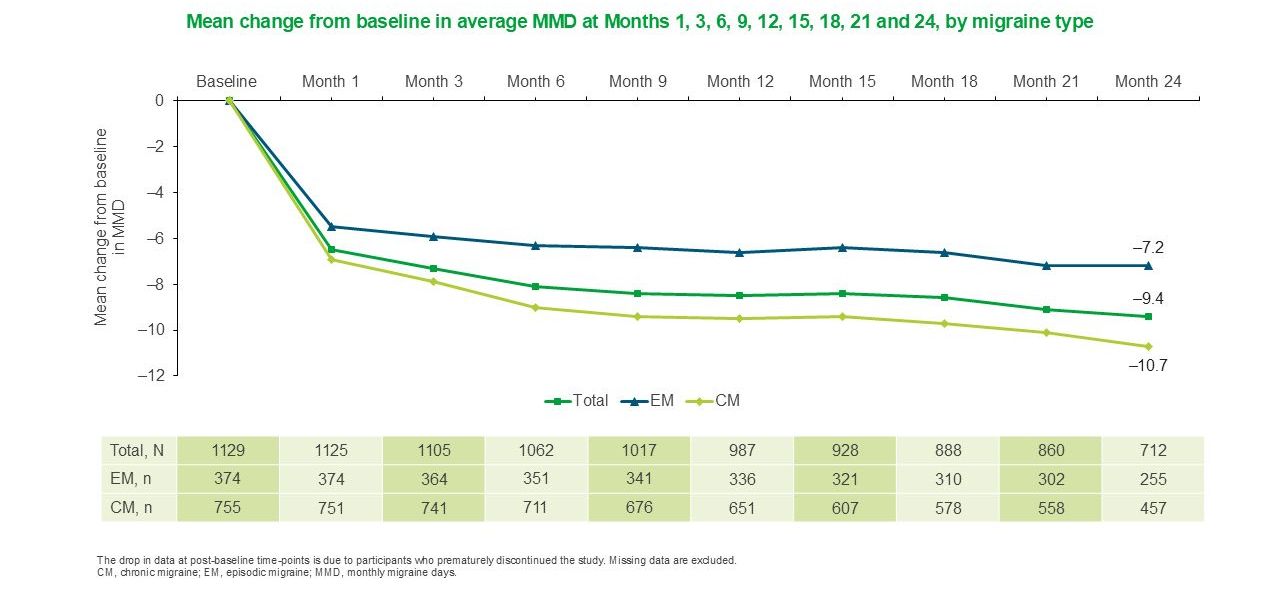
In the Phase IV Pan-European Real Life (PEARL) study, 56.5% (637/1,128) of patients achieved ≥50% reduction in MMDs during the six months after AJOVY® initiation (primary endpoint).8
In EM patients, 66.3% (248/374) achieved ≥50% reduction in MMDs during the six months after AJOVY® initiation.8
In CM patients, 51.6% (389/754) achieved ≥50% reduction in MMDs during the six months after AJOVY® initiation.8
For both EM patients and CM patients in the PEARL study, mean changes from baseline in MMD were sustained from months 1 to 248
AJOVY® safety
AJOVY® has an established safety and tolerability profile.¹ The most commonly reported adverse events are local reactions at the injection site (pain, induration, erythema and pruritus)1
In the Phase III HALO long-term extension study (n=1890), AJOVY® demonstrated a low rate of treatment discontinuation due to tolerability concerns (4%) and a lack of efficacy (4%)†15

No new safety signals were observed with AJOVY® in the real-world study, PEARL, over a 24-month follow-up period8
The most common AJOVY® related AEs, in the real-world PEARL study, were general disorders & administration site conditions (22.2%; 253/1140) and gastrointestinal disorders (6.9%; 79/1140)8
Resources

Getting to know AJOVY®

RFD Patient awareness